
Happy Birthday, Rob! Team Blue-White® Celebrates Our President & CEO
Happy birthday Rob Gledhill Team Blue-White — Blue-White company news celebration highlighting Blue-White CEO Rob Gledhill birthday and leadership culture
Chlorine and chlorine compounds, like sodium hypochlorite, continue to be the go-to chemicals for disinfection in both pretreatment and post-treatment applications. But use of chlorine has some drawbacks. For example, chlorine reacts with organic matter to form disinfection byproducts (DBPs). Chlorine also breaks down quickly, making long-term storage difficult. Chlorinated water that enters the environment may be harmful to plants, insects, and animals.
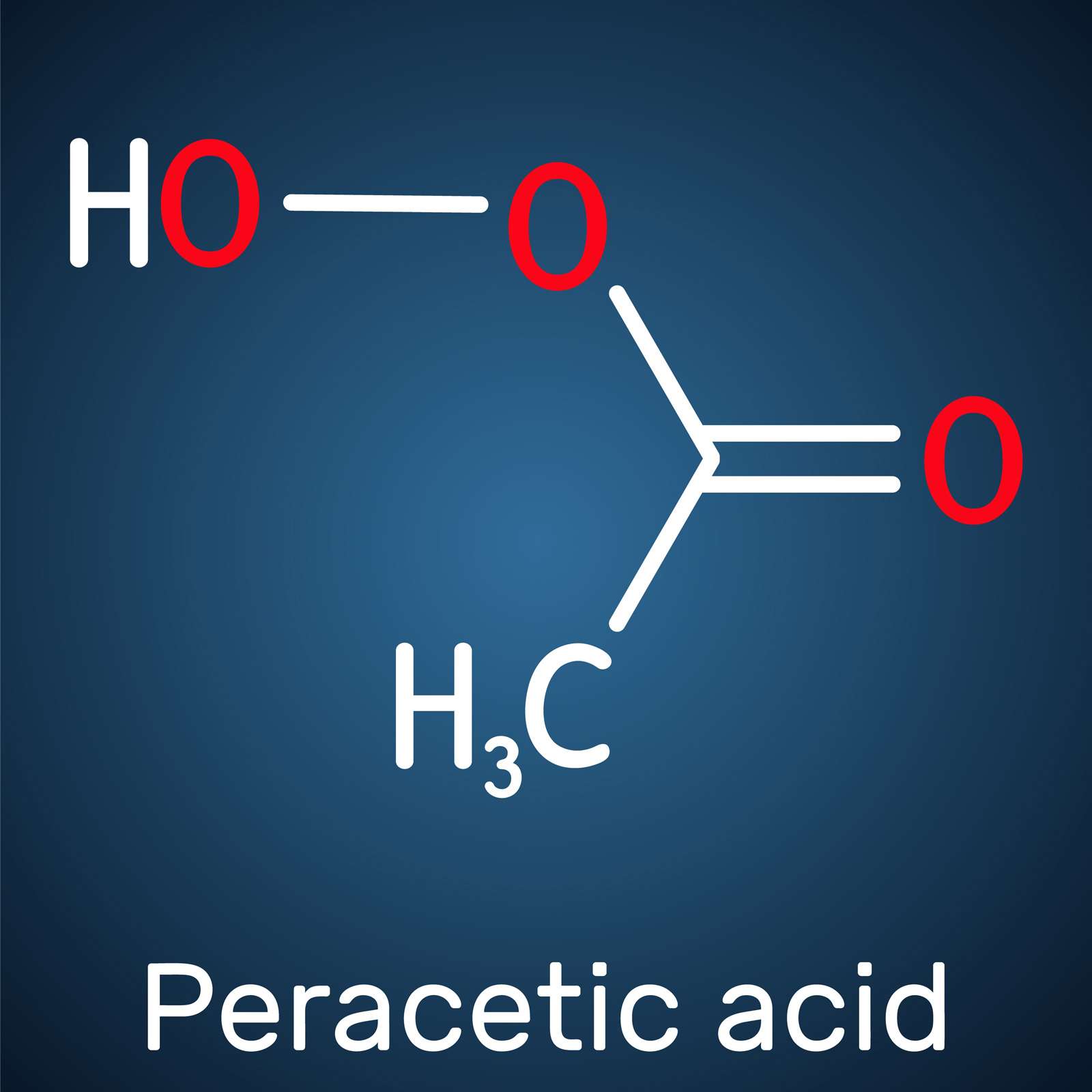
This has led to a growing interest in peracetic acid (PAA) as an alternative to chlorine for disinfection in many applications. PAA has a unique chemistry that is highly effective against bacteria and other contaminants. It does not form any known DBPs and has less environmental impact. However, before substituting chlorine with PAA, it is critical to understand the chemistry, its applications, and methods of handling and dosing.
PAA is an organic chemical made by combining acetic acid with hydrogen peroxide using a catalyst. It has a vinegar-like odor and is typically sold in a solution of acetic acid, peroxide, and water. PAA was first used as a disinfectant in the early 1900s and was registered as a disinfectant with the U.S. EPA in 1985. Benefits of PAA include:
Environmentally friendly. When it oxidizes, PAA breaks down into acetic acid, water, and oxygen. This makes it safer to use on effluent that is discharged into source water or other bodies with high levels of organic content. PAA oxidizes almost instantly, so there is rarely any residual chemical after discharge.
Regulation agnostic. For now, there are few regulations that affect the use of PAA, as it breaks down quickly and does not form any known DBPs.
Long shelf life. PAA has an estimated shelf life of six months to two years. By comparison, sodium hypochlorite has a shelf life of about 90 days.
Of course, PAA is not without its drawbacks. Some things to note about the substance include:
Highly corrosive. PAA can cause damage to the skin, eyes, and lungs. Those handling the chemical should do so in a wellventilated room while wearing appropriate protective gear. It can also corrode metal and some polymers.
Limited effectiveness on viruses. While PAA instantly kills bacteria and fungi, it has been proven less effective on protozoa and viruses, including Giardia and Cryptosporidium. (It will kill SARS-CoV-2 on hard surfaces.)
Higher cost, lower supply. For now, PAA is more expensive than chlorine-based disinfectants and the number of manufacturers and distributors is far fewer. However, as acceptance of the substance increases, these factors are likely to change.

At present, the two most common applications for PAA are in healthcare and food sanitation, due to its ability to break down into predominantly neutral components (oxygen and water).
That being said, PAA has been finding increased usage in industrial wastewater treatment applications. Manufacturers that produce organic waste, such as food processors and breweries, are increasingly applying PAA as a pretreatment before further treating and discharging effluent.
A number of studies have also been conducted regarding the effectiveness of PAA in municipal wastewater treatment to reduce biofouling. So far, it is finding adoption among municipalities with high levels of industrial waste for which UV treatment has been ineffective. It is also popular in areas that are struggling to meet state or federal maximums for chlorine-derived DBPs.
In addition, PAA is beginning to make inroads in drinking water treatment. Its main use is to treat influents to remove bacteria and oxidize other microorganic matter. Used in this manner, it is not a substitute for chlorine, but rather a way of mitigating total organic matter that can react with chlorine to form DBPs downstream.
Dosing PAA into a water treatment system requires the right equipment. The caustic properties make it difficult to run through some kinds of pumps. For high-flow applications, a peristaltic pump may work best, although it’s important to ensure that the pump tube has good chemical resistance. PAA does off-gas which can cause vapor locking in diaphragm pumps, however, peristaltic pumps will not be affected.
Multi-diaphragm pumps overcome the vapor lock problem. Blue- White’s exclusive hyperdrive technology ensures the Multi Diaphragm CHEM-FEED® MD3, MD1, CD3 and CD1 will deliver smooth, precise and dependable chemical feed with no vapor lock and no lost prime. These pumps are quick to install, easy to operate and require minimal maintenance.
Written by:
Blue-White® Industries
714-893-8529

Happy birthday Rob Gledhill Team Blue-White — Blue-White company news celebration highlighting Blue-White CEO Rob Gledhill birthday and leadership culture

Discover the benefits of integrating chemical metering pumps into SCADA for real-time monitoring, improved dosing accuracy, compliance, and reduced labor costs

Happy Cinco de Mayo team celebration from Blue-White company news—Team Blue-White pastries Porto’s Bakery, corporate holiday message from Blue-White®

Blue-White team meet at IFAT 2026 Munich water wastewater solutions in Hall 2 Booth B2.200 Chat with Steve Soroko and others for top quality solutions
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-advertisement | 1 year | Set by the GDPR Cookie Consent plugin, this cookie is used to record the user consent for the cookies in the "Advertisement" category . |
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| CookieLawInfoConsent | 1 year | Records the default button state of the corresponding category & the status of CCPA. It works only in coordination with the primary cookie. |
| elementor | never | This cookie is used by the website. It allows the website owner to implement or change the website's content in real-time. |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |
| Cookie | Duration | Description |
|---|---|---|
| _ga | 2 years | The _ga cookie, installed by Google Analytics, calculates visitor, session and campaign data and also keeps track of site usage for the site's analytics report. The cookie stores information anonymously and assigns a randomly generated number to recognize unique visitors. |
| _gat_gtag_UA_85334924_1 | 1 minute | Set by Google to distinguish users. |
| _gid | 1 day | Installed by Google Analytics, _gid cookie stores information on how visitors use a website, while also creating an analytics report of the website's performance. Some of the data that are collected include the number of visitors, their source, and the pages they visit anonymously. |
| CONSENT | 2 years | YouTube sets this cookie via embedded youtube-videos and registers anonymous statistical data. |
| Cookie | Duration | Description |
|---|---|---|
| VISITOR_INFO1_LIVE | 5 months 27 days | A cookie set by YouTube to measure bandwidth that determines whether the user gets the new or old player interface. |
| YSC | session | YSC cookie is set by Youtube and is used to track the views of embedded videos on Youtube pages. |
| yt-remote-connected-devices | never | YouTube sets this cookie to store the video preferences of the user using embedded YouTube video. |
| yt-remote-device-id | never | YouTube sets this cookie to store the video preferences of the user using embedded YouTube video. |
Please fill out the form to request a quote.
A sales rep will reach out to you.
| Image | Catalog Number | Description | Price | Buy |
|---|